UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
| QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For
the quarterly period ended
OR
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission
file number:
(Exact name of registrant as specified in its charter)
| (State or Other Jurisdiction of | (I.R.S. Employer | |
| Incorporation or Organization) | Identification No.) | |
| (Address of Principal Executive Office) | (Zip Code) |
Registrant’s
telephone number, including area code:
Indicate
by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2)
has been subject to such filing requirements for the past 90 days.
Indicate
by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule
405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post
such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| ☒ | Smaller reporting company | ||
| Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Trading Symbol(s) | Name of each exchange on which registered | ||
The
(The Nasdaq Capital Market) |
As of May 15, 2026, the number of outstanding shares of the registrant’s common stock, par value $ per share, was approximately .
COCRYSTAL PHARMA, INC.
FORM 10-Q FOR THE QUARTER ENDED MARCH 31, 2026
INDEX
| 2 |
Part I – FINANCIAL INFORMATION
COCRYSTAL PHARMA, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
(in thousands, except per share data)
| March 31, 2026 | December 31, 2025 | |||||||
| (unaudited) | ||||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash | $ | $ | ||||||
| Restricted cash | ||||||||
| Grant receivable | ||||||||
| Tax credit receivable | ||||||||
| Prepaid expenses and other current assets | ||||||||
| Total current assets | ||||||||
| Property and equipment, net | ||||||||
| Deposits | ||||||||
| Operating lease right-of-use
assets, net (including $ | ||||||||
| Total assets | $ | $ | ||||||
| Liabilities and stockholders’ equity | ||||||||
| Current liabilities: | ||||||||
| Accounts payable and accrued expenses | $ | $ | ||||||
| Current
maturities of operating lease liabilities (including $ | ||||||||
| Total current liabilities | ||||||||
| Long-term liabilities: | ||||||||
| Operating
lease liabilities (including $ | ||||||||
| Total long-term liabilities | ||||||||
| Total liabilities | ||||||||
| Commitments and contingencies | ||||||||
| Stockholders’ equity: | ||||||||
| Common stock, $ a par value: shares authorized as of March 31, 2026 and December 31, 2025; and shares issued and outstanding as of March 31, 2026 and December 31, 2025 | ||||||||
| Additional paid-in capital | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total stockholders’ equity | ||||||||
| Total liabilities and stockholders’ equity | $ | $ | ||||||
See accompanying notes to condensed consolidated financial statements.
| F-1 |
COCRYSTAL PHARMA, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS
(unaudited)
(in thousands, except per share data)
| Three months ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Revenues and grant income: | ||||||||
| Grant income | ||||||||
| Operating expenses: | ||||||||
| Research and development | ||||||||
| General and administrative | ||||||||
| Total operating expenses | ||||||||
| Loss from operations | ( | ) | ( | ) | ||||
| Other income: | ||||||||
| Interest income, net | ||||||||
| Foreign exchange gain, net | ||||||||
| Total other income, net | ||||||||
| Net loss | $ | ( | ) | $ | ( | ) | ||
| Net loss per common share, basic and diluted | $ | ) | $ | ) | ||||
| Weighted average number of common shares, basic and diluted | ||||||||
See accompanying notes to condensed consolidated financial statements.
| F-2 |
COCRYSTAL PHARMA, INC.
CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
For the three months ended March 31, 2026 and 2025
(unaudited)
(in thousands)
| Common Stock | Additional
Paid-in | Accumulated | Total Stockholders’ | |||||||||||||||||
| Shares | Amount | Capital | Deficit | Equity | ||||||||||||||||
| Balance as of December 31, 2025 | $ | $ | $ | ( | ) | $ | | |||||||||||||
| Stock-based compensation | - | |||||||||||||||||||
| Shares issued from RSU Award | ||||||||||||||||||||
| Net loss | - | ( | ) | ( | ) | |||||||||||||||
| Balance as of March 31, 2026 | $ | $ | $ | ( | ) | $ | ||||||||||||||
| Common Stock | Additional
Paid-in | Accumulated | Total Stockholders’ | |||||||||||||||||
| Shares | Amount | Capital | Deficit | Equity | ||||||||||||||||
| Balance as of December 31, 2024 | $ | $ | $ | ( | ) | $ | | |||||||||||||
| Stock-based compensation | - | |||||||||||||||||||
| Net loss | - | ( | ) | ( | ) | |||||||||||||||
| Balance as of March 31, 2025 | $ | $ | $ | ( | ) | $ | ||||||||||||||
See accompanying notes to condensed consolidated financial statements.
| F-3 |
COCRYSTAL PHARMA, INC.
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(unaudited)
(in thousands)
| Three months ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Operating activities: | ||||||||
| Net loss | $ | ( | ) | $ | ( | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation and amortization expense | ||||||||
| Stock-based compensation | ||||||||
| Decrease right of use assets | ||||||||
| Changes in operating assets and liabilities: | ||||||||
| Grant receivable | ( | ) | ||||||
| Prepaid expenses and other current assets | ( | ) | ||||||
| Tax credit receivable | ( | ) | ||||||
| Deposits | ( | ) | ||||||
| Accounts payable and accrued expenses | ( | ) | ||||||
| Operating lease liabilities | ( | ) | ( | ) | ||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| Net decrease in cash and restricted cash | ( | ) | ( | ) | ||||
| Cash and restricted cash at beginning of period | ||||||||
| Cash and restricted cash at end of period | $ | $ | ||||||
See accompanying notes to condensed consolidated financial statements.
| F-4 |
COCRYSTAL PHARMA, INC.
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
FOR THE THREE MONTHS ENDED MARCH 31, 2026 AND 2025
(unaudited)
1. Organization and Business
Cocrystal Pharma, Inc. (“we”, the “Company” or “Cocrystal”), a clinical stage biopharmaceutical company incorporated in Delaware, has been developing novel technologies and approaches to create first-in-class or best-in-class antiviral drug candidates. Our focus is to pursue the development and commercialization of broad-spectrum antiviral drug candidates that will transform the treatment and prophylaxis of viral diseases in humans. By concentrating our research and development efforts on viral replication inhibitors, we plan to leverage our infrastructure and expertise in these areas.
The Company’s activities since inception have principally consisted of acquiring product and technology rights, raising capital, and performing research and development. Successful completion of the Company’s development programs, obtaining regulatory approvals of its products and, ultimately, the attainment of profitable operations is dependent on future events, including, among other things, its ability to access potential markets, secure financing, develop a customer base, attract, retain and motivate qualified personnel, and develop strategic alliances.
Liquidity and going concern
The
Company’s consolidated financial statements are prepared using generally accepted accounting principles in the United States of
America applicable to a going concern, which contemplates the realization of assets and the satisfaction of liabilities in the normal
course of business. The Company has incurred net losses and negative operating cash flows since inception. For the three months ended
March 31, 2026, the Company recorded a net loss of approximately $
On
March 31, 2026, the Company had cash and restricted cash of approximately $
The Company’s activities since inception have principally consisted of acquiring product and technology rights, raising capital, and performing research and development. Successful completion of the Company’s development programs, obtaining regulatory approvals of its products and, ultimately, the attainment of profitable operations is dependent on future events, including, among other things, its ability to develop, obtain approval and commercialize antiviral drug candidates, including access potential markets, secure financing, develop a customer base, and to attract, retain and motivate qualified personnel, and develop strategic alliances. Through March 31, 2026, the Company has primarily funded its operations through equity offerings and limited revenue.
| F-5 |
The Company will need to continue obtaining adequate capital to fund operating losses until it becomes profitable. The Company can give no assurances that the additional capital it is able to raise, if any, will be sufficient to meet its needs, or that any such financing will be obtainable on acceptable terms. Our future cash requirements, and the timing of those requirements, will depend on a number of factors, including economic conditions, the approval and success of our products in development, the continued progress of research and development of our product candidates, the timing and outcome of clinical trials and regulatory approvals, the costs involved in preparing, filing, prosecuting, maintaining, defending, and enforcing patent claims and other intellectual property rights, the status of competitive products, the availability of financing, our success in developing markets for our product candidates and legal proceedings that may arise. We have historically not generated sustained positive cash flow and if we are not able to secure additional funding when needed, we may have to delay, reduce the scope of, or eliminate one or more of our clinical trials or research and development programs. If the Company is unable to obtain adequate capital, it could be forced to cease operations or substantially curtail its drug development activities. The Company expects to continue incurring substantial operating losses and negative cash flows from operations over the next several years during its pre-clinical and clinical development phases.
2. Basis of Presentation and Significant Accounting Policies
Basis of Presentation
The accompanying condensed consolidated financial statements have been prepared in accordance with United States generally accepted accounting principles (“U.S. GAAP”) for interim financial information, the instructions to Form 10-Q and Article 10 of Regulation S-X set forth by the Securities and Exchange Commission (“SEC”). They do not include all of the information and notes required by U.S. GAAP for complete financial statements. In the opinion of management, all adjustments (consisting of normal recurring accruals) considered necessary for a fair presentation have been included. The results of operations for the interim periods presented are unaudited and are not necessarily indicative of the results of operations for the entire fiscal year. For further information, refer to the consolidated financial statements and footnotes thereto included in the Company’s annual report on Form 10-K for the year ended December 31, 2025 filed on March 31, 2026 (“Annual Report”).
Principles of Consolidation
The consolidated financial statements include the accounts of Cocrystal Pharma, Inc. and its wholly owned subsidiaries: Cocrystal Discovery, Inc., Cocrystal Pharma Australia Pty Ltd. (“Cocrystal Australia”), RFS Pharma, LLC and Cocrystal Merger Sub, Inc. Intercompany transactions and balances have been eliminated. Cocrystal Discovery, Inc. conducts all of the Company’s research and development activities and oversees ongoing clinical trials conducted by others. Cocrystal Australia operates clinical trials in Australia. The other two subsidiaries are inactive.
Segments
The Company’s Co-Chief Executive Officer and President (“CEO”) is our chief operating decision maker (“CODM”) and evaluates performance and makes operating decisions about allocating resources based on financial data presented on a consolidated basis. Because our CODM evaluates financial performance on a consolidated basis, the Company has determined that it operates as a single reportable segment composed of the consolidated financial results of Cocrystal Pharma, Inc. The measure of segment assets is reported on the consolidated balance sheets as total assets (see Note 9).
| F-6 |
Use of Estimates
Preparation of the Company’s consolidated financial statements in conformance with U.S. GAAP requires the Company’s management to make estimates and assumptions that impact the reported amounts of assets, liabilities, revenues and expenses, and the disclosure of contingent assets and liabilities in the Company’s consolidated financial statements and accompanying notes.
The most significant estimates in the Company’s consolidated financial statements relate to clinical trial costs and accruals and the fair value of stock-based compensation. The Company bases estimates and assumptions on historical experience, when available, and on various factors that it believes to be reasonable under the circumstances. The Company evaluates its estimates and assumptions on an ongoing basis, and its actual results may differ from estimates made under different assumptions or conditions.
Concentrations of Credit Risk
Financial
instruments that potentially subject the Company to significant concentrations of credit risk consist primarily of cash deposited in
accounts held at two U.S. financial institutions, which may, at times, exceed federally insured limits of $
Risks and uncertainties
The Company’s future results of operations involve a number of risks and uncertainties. Factors that could affect the Company’s future operating results and cause actual results to vary materially from expectations include, but are not limited to, rapid technological change, ability to obtain regulatory approvals, competition from currently available treatments and therapies, competition from larger companies, effective protection of proprietary technology, maintenance of strategic relationships, and dependence on key individuals.
Products developed by the Company will require clearances from the U.S. Food and Drug Administration (the “FDA”) and other international regulatory agencies prior to commercial sales in their respective markets. The Company’s products may not receive the necessary clearances and if they are denied clearance, clearance is delayed, or the Company is unable to maintain clearance, the Company’s business could be materially, adversely impacted.
See Item 1A- Risk Factors in our Annual Report on Form 10-K for the year ended December 31, 2025 for more information on the risks and uncertainties we face.
Foreign Currency Transactions
The Company and its subsidiaries use the U.S. dollar as functional currency. Foreign currency transactions are initially measured and recorded in the functional currency using the exchange rate on the date of the transaction. Foreign exchange gains and losses arising from settlement of foreign currency transactions are recognized in profit and loss.
Cocrystal Australia maintains its records in Australian dollars. The monetary assets and liabilities of Cocrystal Australia are remeasured into the functional currency using the closing rate at the end of every reporting period. All nonmonetary assets and liabilities and related profit and loss accounts are remeasured into the functional currency using the historical exchange rates. Profit and loss accounts, other than those that are remeasured using the historical exchange rates, are remeasured into the functional currency using the average exchange rate for the period. Foreign exchange gains and losses arising from the remeasurement into the functional currency is recognized in profit and loss.
Fair Value Measurements
FASB Accounting Standards Codification (“ASC”) 820 defines fair value, establishes a framework for measuring fair value under U.S. GAAP and enhances disclosures about fair value measurements. Fair value is defined under ASC 820 as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value under ASC 820 must maximize the use of observable inputs and minimize the use of unobservable inputs. The standard describes a fair value hierarchy based on three levels of inputs, of which the first two are considered observable and the last unobservable, that may be used to measure fair value which are the following:
| Level 1 — quoted prices in active markets for identical assets or liabilities. |
| F-7 |
| Level 2 — other significant observable inputs for the assets or liabilities through corroboration with market data at the measurement date. | |
| Level 3 — significant unobservable inputs that reflect management’s best estimate of what market participants would use to price the assets or liabilities at the measurement date. |
At March 31, 2026 and December 31, 2025, the carrying amounts of financial assets and liabilities, such as cash, grant receivable, tax receivable, other assets, and accounts payable and accrued expenses approximate their fair values due to their short-term nature. The carrying values of leases payable approximate their fair values due to the fact that the interest rates on these obligations are based on prevailing market interest rates.
Long-Lived Assets
The Company regularly reviews the carrying value and estimated lives of its long-lived assets, including property and equipment, to determine whether indicators of impairment may exist which warrant adjustments to carrying values or estimated useful lives. The determinants used for this evaluation include management’s estimate of the asset’s ability to generate positive income from operations and positive cash flow in future periods as well as the strategic significance of the assets to the Company’s business objective. Should an impairment exist, the impairment loss would be measured based on the excess of the carrying amount over the asset’s fair value.
Research and Development Expenses
Research and development costs consist primarily of fees paid to consultants and outside service providers, and other expenses relating to the acquisition, design, development and testing of the Company’s clinical products. All research and development costs are expensed as incurred. Research and development costs are presented net of tax credits.
The
Company’s Australian subsidiary is entitled to receive government assistance in the form of refundable and non-refundable research
and development tax credits (“Refundable Tax Credits”) from the federal and provincial taxation authorities, based on qualifying
expenditures incurred during the fiscal year. The Refundable Tax Credits are from the provincial taxation authorities and are not dependent
on its ongoing tax status or tax position and accordingly are not considered part of income taxes. The Company records Refundable Tax
Credits as a reduction of research and development expenses when the Company can reasonably estimate the amounts and it is more likely
than not, they will be received. As of March 31, 2026, the balance of Refundable Tax Credits was approximately $
Income Taxes
The Company accounts for income taxes under the asset and liability method. Under this method, deferred tax assets and liabilities are determined based on differences between financial reporting and tax bases of assets and liabilities and are measured using enacted tax rates and laws that are expected to be in effect when the differences are expected to be recovered or settled. Realization of deferred tax assets is dependent upon future taxable income. A valuation allowance is recognized if it is more likely than not that some portion or all of a deferred tax asset will not be realized based on the weight of available evidence, including expected future earnings. The Company recognizes an uncertain tax position in its financial statements when it concludes that a tax position is more likely than not to be sustained upon examination based solely on its technical merits. Only after a tax position passes the first step of recognition will measurement be required. Under the measurement step, the tax benefit is measured as the largest amount of benefit that is more likely than not to be realized upon effective settlement. This is determined on a cumulative probability basis. The full impact of any change in recognition or measurement is reflected in the period in which such change occurs. The Company elects to accrue any interest or penalties related to income taxes as part of its income tax expense.
| F-8 |
As
of March 31, 2026, the Company assessed its income tax expense based on its projected future taxable income for the year ending December
31, 2025 and therefore recorded
The Company recognizes compensation expense using a fair value-based method for costs related to stock-based payments, including stock options. The fair value of options awarded to employees is measured on the date of grant using the Black-Scholes option pricing model and is recognized as expense over the requisite service period on a straight-line basis. Recognition of compensation expense for non-employees is in the same period and manner as if the Company had paid cash for the services.
Use of the Black-Scholes option pricing model requires the input of subjective assumptions including expected volatility, expected term, and a risk-free interest rate. The Company estimates volatility using a blend of its own historical stock price volatility as well as that of market comparable entities since the Company’s common stock has limited trading history and limited observable volatility of its own. The expected term of the options is estimated by using the SEC Staff Bulletin No. 107’s Simplified Method for Estimate Expected Term. The risk-free interest rate is estimated using comparable published federal funds rates.
Grant Income
The Company accounts for government grant funding by analogy to ASC 958-605 because U.S. GAAP does not contain specific guidance for business entities receiving government grants.
Grant income is recognized as the Company incurs qualifying research and development expenditures and satisfies the conditions associated with the grant arrangement.
In
October 2025, the Company announced that it had received a $
During
the three months ended March 31, 2026, the Company recognized grant income of $
The Company accounts for and discloses net income (loss) per common share in accordance with FASB ASC Topic 260, Earnings Per Share. Basic income (loss) per common share is computed by dividing income (loss) attributable to common stockholders by the weighted average number of common shares outstanding. Diluted net income (loss) per common share is computed by dividing net income (loss) attributable to common stockholders by the weighted average number of common shares that would have been outstanding during the period assuming the issuance of common stock for all potential dilutive common shares outstanding. Potential common shares consist of shares issuable upon the exercise of stock options and restricted stock units.
| March 31, | ||||||||
| 2026 | 2025 | |||||||
| Outstanding options to purchase common stock | ||||||||
| Warrants to purchase common stock | ||||||||
| Unvested restricted stock units | ||||||||
| Total | ||||||||
Recent Accounting Pronouncements
In November 2024, the Financial Accounting Standards Board (FASB) issued ASU No. 2024-03, Income Statement—Reporting Comprehensive Income—Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses which includes amendments that require disclosure in the notes to financial statements of specified information about certain costs and expenses, including purchases of inventory; employee compensation; and depreciation, amortization and depletion expenses for each caption on the income statement where such expenses are included. The amendments are effective for the Company’s annual periods beginning January 1, 2027, with early adoption permitted, and should be applied either prospectively or retrospectively. The Company is in the process of evaluating this ASU to determine its impact on the Company’s disclosures.
Other authoritative guidance issued by the FASB (including technical corrections to the ASC), the American Institute of Certified Public Accountants, and the SEC did not, or are not expected to, have a material impact on the Company’s consolidated financial statements and related disclosures.
| F-9 |
3. Property and Equipment
Property
and equipment are recorded at cost and depreciated over the estimated useful lives of the underlying assets (three to
| March 31, 2026 | December 31, 2025 | |||||||
| Lab equipment (excluding equipment under finance leases) | $ | $ | ||||||
| Finance lease right-of-use lab equipment obtained in exchange for finance lease liabilities, net | ||||||||
| Computer and office equipment | ||||||||
| Total property and equipment | ||||||||
| Less: accumulated depreciation and amortization | ( | ) | ( | ) | ||||
| Property and equipment, net | $ | $ | ||||||
Total
depreciation and amortization expense were approximately $
4. Accounts Payable and Accrued Expenses
Accounts payable and accrued expenses consisted of the following (in thousands) as of:
| March 31, 2026 | December 31, 2025 | |||||||
| Accounts payable | $ | $ | ||||||
| Accrued compensation | ||||||||
| Accrued other expenses | ||||||||
| Total accounts payable and accrued expenses | $ | $ | ||||||
Accounts payable and accrued expenses contain unpaid general and administrative expenses and costs related to research and development that have been billed and estimated unbilled, respectively, as of period-end.
5. Common Stock and Warrants
As of March 31, 2026, the Company has authorized shares of common stock, $ par value per share. The Company had and shares issued and outstanding as of March 31, 2026 and December 31, 2025.
On
September 12, 2025, the Company, entered into a securities purchase agreement with certain accredited investors, pursuant to which the
Company sold to the investors (i) in a registered direct offering, an aggregate of shares of the Company’s
common stock, at a price of $ per share and (ii) in a concurrent private placement, warrants to purchase up to an aggregate
of
Warrant Activity Table:
| Total Options Outstanding | Weighted Average Exercise Price | Aggregate Intrinsic Value | ||||||||||
| Balance at December 31, 2025 | $ | $ | ||||||||||
| Granted | - | |||||||||||
| Exercised | - | |||||||||||
| Forfeited | - | |||||||||||
| Outstanding at March 31, 2026 | $ | $ | ||||||||||
Equity Incentive Plans
The Company adopted an equity incentive plan in 2015 (the “2015 Plan”) under which shares of common stock have been reserved for issuance to employees, and non-employee directors and consultants of the Company. Recipients of incentive stock options granted under the 2015 Plan shall be eligible to purchase shares of the Company’s common stock at an exercise price equal to no less than the estimated fair market value of such stock on the date of grant. The maximum term of options granted under the 2015 Plan is . On June 16, 2021, the Company’s stockholders voted to approve an amendment to the 2015 Plan to increase the number of shares of common stock authorized for issuance under the 2015 Plan from to shares. As of March 31, 2026, no shares remained available for future grants under the 2015 Plan. The 2015 Plan expired on June 29, 2025.
| F-10 |
On April 2, 2025, the Board of Directors of the Company approved and adopted an Equity Incentive Plan (the “2025 Plan”), which has an effective date of March 31, 2025. On June 25, 2025, the 2025 Plan was approved by our stockholders at our annual meeting of stockholders. The 2025 Plan provides for the grant of incentive stock options, qualified stock options, restricted stock awards, restricted stock units, stock appreciation rights, and performance shares or units and cash awards. Awards may be granted under the 2025 Plan to our employees, directors and independent contractors. the aggregate number of shares of Common Stock which shall be available for grants or payments of Awards under the 2025 Plan during its term shall initially be (the “Total Plan Shares”). The Total Plan Shares will automatically increase on January 1st of each year, for a period of nine years commencing on January 1, 2026, in an amount equal to % of the total number of shares of Common Stock outstanding as of December 31 of the preceding calendar year on a fully diluted basis.
The 2025 Plan also provides that, notwithstanding the annual increase provision, in no event will the increase in Total Plan Shares available under the 2025 Plan pursuant to the increase provision exceed additional shares (or a total of up to Total Plan Shares), subject to adjustment as provided under the 2025 Plan.
As of March 31, 2026, shares remained available for future grants under the 2025 Plan.
Common Stock Reserved for Future Issuance
The following table presents information concerning common stock available for future issuance (in thousands) as of March 31, 2026:
| Shares
Available for Grant | ||||
| Stock options issued and outstanding | ||||
| Restricted stock units issued and outstanding | ||||
| Shares authorized for future option grants | ||||
| Warrants outstanding | ||||
| Balance at March 31, 2026 | $ | |||
Stock Options
| Total
Options Outstanding | Weighted
Average Exercise Price | Aggregate Intrinsic Value | ||||||||||
| Balance at December 31, 2025 | $ | $ | ||||||||||
| Exercised | - | |||||||||||
| Granted | - | |||||||||||
| Cancelled | ( | ) | - | |||||||||
| Balance at March 31, 2026 | $ | $ | ||||||||||
In January 2026, the Compensation Committee of the Company’s Board of Directors granted a total of stock options with a fair value of $ effective as of January 9, 2026. The Company granted stock options to directors, executives, employees, and consultants. The options are ten-year incentive stock options exercisable at $ per share and vesting as follows: one-half vest on the one-year anniversary of the grant date and the remainder vest in eight equal quarterly instalments commencing on March 31, 2027.
| Three months ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Risk-Free interest rate | % | % | ||||||
| Expected dividend yield | % | % | ||||||
| Expected volatility | ||||||||
| Expected term (in years) | - | |||||||
Restricted Stock Units
On August 12, 2024, the Company’s Compensation Committee approved the issuance of restricted stock unit (“RSU”) awards to non-employee directors, officers, consultants and employees. The aggregate fair value of the restricted stock unit awards granted was estimated to be $ using the market price of the stock on the date of the grant which is expensed using the straight-line method over the vesting period.
| Total
Restricted Stock Units Outstanding | Weighted
Average Fair Value | Aggregate
Intrinsic Value | ||||||||||
| Unvested December 31, 2025 | $ | $ | ||||||||||
| Granted | - | |||||||||||
| Forfeited | ( | ) | - | |||||||||
| Vested | ( | ) | - | |||||||||
| Unvested and expected to vest at March 31, 2026 | $ | $ | ||||||||||
| F-11 |
The Company accounts for share-based awards to employees and nonemployee directors and consultants in accordance with the provisions of ASC 718, Compensation—Stock Compensation., and under the recently issued guidance following FASB’s pronouncement, ASU 2018-07, Compensation—Stock Compensation (Topic 718): Improvements to Nonemployee Share-Based Payment Accounting. Under ASC 718, and applicable updates adopted, share-based awards are valued at fair value on the date of grant and that fair value is recognized over the requisite service, or vesting, period. The Company values its equity awards using the Black-Scholes option pricing model, and accounts for forfeitures when they occur. For the three months ended March 31, 2026 and 2025, equity-based compensation expense recorded was approximately $ and $, respectively.
As of March 31, 2026, there was approximately $ of total unrecognized compensation expense related to non-vested stock options that is expected to be recognized over a weighted average period of years. For options granted and outstanding, there were options outstanding which were fully vested or expected to vest, with an aggregate intrinsic value of $ , a weighted average exercise price of $ and weighted average remaining contractual term of years at March 31, 2026. For vested and exercisable options, outstanding shares totaled , with an aggregate intrinsic value of $ . These options had a weighted average exercise price of $ per share and a weighted-average remaining contractual term of years at March 31, 2026.
The aggregate intrinsic value of outstanding and exercisable options at March 31, 2026 was calculated based on the closing price of the Company’s common stock as reported on The Nasdaq Capital Market on March 31, 2026 of $ per share less the exercise price of the options. The aggregate intrinsic value is calculated based on the positive difference between the closing fair market value of the Company’s common stock and the exercise price of the underlying options.
7. Commitments and Contingencies
Commitments
In the ordinary course of business, the Company enters into non-cancellable leases to purchase equipment and for its facilities, including related party leases (see Note 8 – Transactions with Related Parties). Leases are accounted for as operating leases or finance leases, in accordance with ASC 842, Leases.
Operating Leases
The
Company leases office space in Miami, Florida and research and development laboratory space in Bothell, Washington under operating leases
that expire on September 30, 2027 and January 31, 2031, respectively. For operating leases, the weighted average discount rate is
The following table summarizes the Company’s maturities of operating lease liabilities, by year and in aggregate, as of March 31, 2026 (table in thousands):
| 2026 (excluding the three months ended March 31, 2026) | $ | |||
| 2027 | ||||
| 2028 | ||||
| 2029 | ||||
| 2030 and thereafter | ||||
| Total operating lease payments | ||||
| Less: present value discount | ( | ) | ||
| Total operating lease liabilities | $ |
| F-12 |
As
of March 31, 2026, the total operating lease liability of $
In
April 2023, the Company renewed its lease for the unit 100 at the Bothell, Washington facility (“Bothell 100”) for an 84-month
(
In
September 2023, following the renewal of the Bothell 100 facility lease, the Company amended the agreement to expand the premises to
include Suite 200 (“Bothell 200 facility”). The lease for the Bothell 200 facility has a 60-month (
In
August 2024, the Company renewed its lease for the Miami, Florida location for a
The
operating lease liabilities summarized above do not include variable common area maintenance (the “CAM”) charges, which are
contractual liabilities under the Company’s Bothell, Washington lease. CAM charges for the Bothell, Washington facility are calculated
annually based on actual common expenses for the building incurred by the lessor and proportionately billed to tenants based on leased
square footage. For the three months ended March 31, 2026 and 2025, approximately $
The lessor of the Miami, Florida lease is a limited liability company controlled by Dr. Phillip Frost, a director and a principal stockholder of the Company.
| F-13 |
For
the three months ended March 31, 2026 and 2025, operating lease expense, including short-term leases, finance leases and CAM charges,
totaled approximately $
Contingencies
From time to time, the Company is a party to, or otherwise involved in, legal proceedings arising in the normal course of business. As of the date of this report, except as described below, the Company is not aware of any proceedings, threatened or pending, against it which, if determined adversely, would have a material effect on its business, results of operations, cash flows or financial position.
8. Transactions with Related Parties
On
August 14, 2024, the Company entered into a three-year lease extension with a limited liability company controlled by Dr. Phillip Frost,
a director and a principal stockholder of the Company. On an annualized basis, straight-line rent expense is approximately $
The
Company paid a lease deposit of $
9. Segment Information
The Company operates and manages its business as one reportable and operating segment dedicated to the research and development of the Company’s novel orally administered antiviral influenza candidate. The measure of segment assets is reported on the balance sheet as total consolidated assets. In addition, the Company manages the business activities on a consolidated basis.
The Company’s CODM reviews financial information presented on a consolidated basis and decides how to allocate resources based on net income (loss).
Significant segment expenses include research and development, salaries, insurance, and stock-based compensation. Operating expenses include all remaining costs necessary to operate our business, which primarily include external professional services and other administrative expenses. The following table presents the significant segment expenses and other segment items regularly reviewed by our CODM (table in thousands):
| Three months ended March 31, | ||||||||
| 2026 | 2025 | |||||||
| Revenue | $ | $ | ||||||
| Less: | ||||||||
| Research and development | ||||||||
| Salaries and personnel costs | ||||||||
| Insurance | ||||||||
| Stock-based compensation | ||||||||
| Operating expenses | ||||||||
| Other income | ( | ) | ( | ) | ||||
| Net loss | $ | $ | ||||||
| F-14 |
ITEM 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Overview
Cocrystal Pharma, Inc. (the “Company” or “Cocrystal”) is a clinical-stage biotechnology company seeking to discover and develop novel antiviral therapeutics as treatments for serious and/or chronic viral diseases. We employ unique structure-based technologies and Nobel Prize winning expertise in efforts to create first- and best-in-class antiviral drugs. These technologies are designed to efficiently deliver small molecule therapeutics that are safe, effective and convenient to administer. We have identified promising preclinical and clinical-stage antiviral compounds for unmet medical needs caused by RNA viruses including norovirus, influenza virus, coronaviruses (including SARS-CoV-2 and MERS-COV), respiratory virus infections and hepatitis C virus (“HCV”).
Impact of Inflation
The Company believes that inflation has not had a material effect on its operations to date, other than the impact of inflation on the general economy. However, there is a risk that the Company’s operating costs could become subject to inflationary pressures in the future particularly based upon United States tariff policy, which could have a material effect on increasing the Company’s operating costs, and which would put additional stress on the Company’s working capital resources.
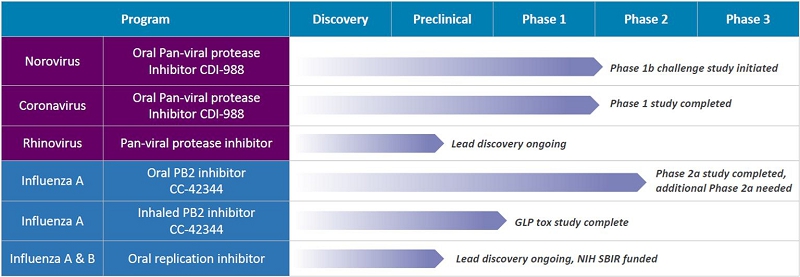
Research and Development Update
During the three months ended March 31, 2026 and more recently the Company continued to focus its research and development efforts primarily in three areas of norovirus, influenza and coronavirus.

Norovirus Program
We developed the novel protease inhibitor CDI-988 as an oral pan-viral treatment of noroviruses and coronaviruses, including SARS-CoV-2 and its variants. CDI-988 was specifically designed and developed using our proprietary structure-based drug discovery platform technology as a broad-spectrum antiviral inhibitor to a highly conserved region in the active site of noroviruses, coronaviruses and other 3CL viral proteases. We believe CDI-988 represents the only oral pan-viral antiviral in development for the treatment and prevention of viral gastroenteritis caused by noroviruses, and coronaviruses, including SARS-CoV-2 and its variants.
Oral CDI-988, the first oral direct-acting protease inhibitor, was clinically evaluated for safety, tolerability and pharmacokinetics including a food-effect cohort in healthy volunteers in a single-center, randomized, double-blind, placebo-controlled Phase 1 study conducted in Australia.
| 3 |
In July 2024 we announced favorable safety and tolerability results from the single-ascending dose (SAD) cohorts of the Phase 1 study with CDI-988. Study participants in the SAD cohorts received CDI-988 in doses ranging from 100 mg to 600 mg. All participants completed the study with no discontinuations. There were no serious adverse events (“SAEs”) or severe treatment-emergent adverse events. No clinically significant observations were noted in laboratory assessments, physical exams or electrocardiograms.
In September 2024 we initiated dosing of the first subjects in the multiple-ascending dose (MAD) portion of the Phase 1 study with CDI-988. Topline Phase 1 study safety and tolerability SAD results and testing of 800 mg for 10 consecutive days were reported in January 2025 indicating favorable safety and tolerability results. We also announced that an additional cohort with a higher dose of 1,200 mg and a shorter treatment duration of five consecutive days would be conducted to further assess CDI-988’s safety, tolerability and pharmacokinetics. In August 2025 we presented favorable safety and tolerability Phase 1 data from all CDI-988 doses, including the high-dose 1200 mg cohort, at the 2025 Military Health System Research Symposium (MHSRS).
In September 2025 we received a Study May Proceed Letter from the FDA to conduct a Phase 1b challenge study in the U.S. evaluating CDI-988 as a norovirus preventive and treatment. In December 2025, we received Institutional Review Board approval from Emory University School of Medicine, the clinical study site for the Phase 1b trial, and announced that subject screening for the study was underway. In February 2026, we announced commencement of the Phase 1b challenge study at Emory University School of Medicine. The study’s primary efficacy endpoint is to assess the reduction in incidence of clinical symptoms, while the secondary efficacy endpoint focuses on the reduction in viral shedding and disease severity. The study will also assess the safety and pharmacokinetic profile of CDI-988.
We recently announced that the FDA has granted Fast Track designation to CDI-988. The FDA Fast Track designation aims to facilitate the development and accelerate the review process for drugs that treat serious conditions and address unmet medical needs and allows for a rolling review of a new drug application (NDA) and might help qualify a product for priority review at the time of NDA submission.
In March 2026, we enrolled the first subjects in our Phase 1b norovirus challenge study to evaluate CDI-988 as both a preventative and treatment for norovirus infections, with the initial cohort evaluating the infectivity rate of the GII.2 (Snow Mountain Virus) challenge inoculum, and subsequent cohorts to be orally administered CDI-988 or placebo.
Influenza Programs
We have several candidates under development for the treatment of influenza infection. CC-42344, a novel PB2 inhibitor, was selected as a preclinical lead as an oral or inhaled treatment of pandemic and seasonal influenza A. This candidate binds to a highly conserved PB2 site of influenza polymerase complex (PB1: PB2: PA) and exhibits a novel mechanism of action. CC-42344 showed excellent in vitro antiviral activity against influenza A strains, including avian pandemic strains and Tamiflu® and Xofluza® resistant strains, and has favorable pharmacokinetic and drug resistance profiles.
In addition to oral candidate of CC-42344, inhaled CC-42344 is being developed for the potential prophylactic treatment of pandemic and seasonal influenza infections. Dry powder inhalation development and toxicology studies have been evaluated.
In December 2023 we received authorization from the United Kingdom Medicines and Healthcare Products Regulatory Agency (MHRA) to conduct a Phase 2a human challenge study with oral CC-42344 as a potential treatment for pandemic and seasonal influenza A. This randomized, double-blind, placebo-controlled study was designed to evaluate the safety, tolerability, viral and clinical measurements of healthy subjects infected with the influenza A virus dosed with oral CC-42344 treatment. While in the Phase 2a study CC-42344 demonstrated favorable safety and tolerability profile and no serious adverse events (“SAEs”) or drug-related discontinuations by study participants, due to unexpectedly low influenza infection among study participants, management determined that the low infectivity obtained in this study hindered antiviral data analysis. A dispute has arisen with the United Kingdom clinical research organization (the “CRO”) that performed the Phase 2a study. The Company contends that the CRO breached its agreement in a number of respects and is requesting that the CRO refund the $6,309,000 it was paid or redo the study. The CRO has implicitly denied liability and is seeking to recover an additional approximately $600,000 from the Company. As of the date of this Report, it appears that the Company will seek to arbitrate the dispute as required under the agreement with the CRO and more recently has filed a demand letter reserving all of our rights and has initiated arbitration. See the risk factor entitled “We face significant risks and uncertainties surrounding our Influenza A program following an initial Phase 2a study which failed to yield scientifically viable results relating to the product candidate’s efficacy” beginning on page 14 of our Annual Report on Form 10-K for the year ended December 31, 2025. Subject to resolution of this issue or our raising capital to conduct another study, we plan to continue development of oral CC-42344 as a treatment for pandemic and seasonal influenza A.
| 4 |
In June 2024 we reported the potential efficacy of CC-42344 against the new Texas avian flu strain from in vitro studies with the recently published genome sequence for H5N1. Using our proprietary structure-based platform technology, the Company reported a high-resolution cocrystal structure of this avian PB2 protein complexed with CC-42344 and confirmed that CC-42344 binds to its highly conserved PB2 region. The in vitro data using purified Texas avian H5N1 PB2 protein further showed in vitro affinity of CC-42344 similar to that of previous data using pandemic avian and seasonal influenza A PB proteins.
We also continue developing novel broad-spectrum influenza antivirals targeting replication enzymes of seasonal and pandemic influenza A and B strains. On October 27, 2025, the Company announced it received a $500,000 Small Business Innovation Research (“SBIR”) Phase I award from the National Institutes of Health (“NIH”) and the National Institute of Allergy and Infectious Diseases (NIAID). The NIH/NIAID Phase I award is designed to assess the scientific, technical and commercial potential of early-stage programs and will support the Company’s development of a novel, oral, broad-spectrum antiviral candidate for the treatment of influenza A and B infections.
Coronavirus Program
As repeated from our norovirus program above, we developed the novel protease inhibitor CDI-988 as an oral pan-viral treatment of noroviruses and coronaviruses, including SARS-CoV-2 and its variants. CDI-988 was specifically designed and developed using our proprietary structure-based drug discovery platform technology as a broad-spectrum antiviral inhibitor to a highly conserved region in the active site of noroviruses, coronaviruses and other 3CL viral proteases.
Oral CDI-988 was clinically evaluated for safety, tolerability and pharmacokinetics including a food-effect cohort in healthy volunteers in a single-center, randomized, double-blind, placebo-controlled Phase 1 study conducted in Australia.
We continue to evaluate CDI-988 for additional program development.
Therapeutic Targets
Influenza: A worldwide public health problem, including the potential for pandemic Avian Flu
Influenza is a severe respiratory illness caused primarily by influenza A or B virus. Influenza A viruses are the only influenza viruses known to cause influenza pandemics. Each year there are approximately 1 billion cases of seasonal influenza worldwide, with 3-5 million severe illnesses and up to 650,000 deaths, according to the World Health Organization (“WHO”). On average about 8% of the U.S. population contracts influenza each season, according to the Centers for Disease Control and Prevention (“CDC”). In addition to the health risk, influenza is responsible for approximately $10.4 billion in direct medical costs in the U.S. annually, according to the National Institutes of Health (“NIH”).
Currently approved antiviral treatments for influenza are effective but burdened with significant viral resistance. Strains of influenza virus resistant to the approved treatments oseltamivir phosphate (Tamiflu®), zanamavir (Relenza®) and baloxavir marboxil (Xofluza®) have appeared and in some cases are predominant. For example, the predominant strain of the 2009 swine influenza pandemic was resistant to oseltamivir. Oseltamivir inhibits influenza neuraminidase enzymes, which are not highly conserved between viral strains. According to the WHO, approximately 16% of the H1N1 isolates circulating worldwide were oseltamivir resistant. Also, treatment-emergent resistance to recently approved baloxavir has been observed during clinical trials and the potential transmission of resistant influenza variants could significantly diminish baloxavir effectiveness.
| 5 |
Norovirus: A worldwide public health problem responsible for close to 90% of the global epidemic, non-bacterial outbreaks of gastroenteritis with no effective treatment or vaccine
Norovirus is a very common and highly contagious virus that causes symptoms of acute gastroenteritis among people of all ages including nausea, vomiting, stomach pain and diarrhea as well as fatigue, fever and dehydration. Norovirus infection can be significantly more severe and prolonged in specific risk groups including infants, children, the elderly and people with immunodeficiency. In immunosuppressed patients, chronic norovirus infection can lead to a debilitating illness with extended periods of nausea, vomiting and diarrhea. Norovirus outbreaks occur most commonly in semi-closed communities and have become notorious for their occurrence in hospitals, nursing homes, childcare facilities, cruise ships, schools, disaster relief sites and military settings.
In the U.S. alone, noroviruses are the leading cause of vomiting and diarrhea from acute gastroenteritis among people of all ages and responsible for an estimated 21 million cases annually, including 109,000 hospitalizations, 465,000 emergency department visits and an estimated 900 deaths, according to the CDC. The NIH estimates the annual burden to the United States at $10.6 billion.
According to the CDC, noroviruses average 685 million cases of acute gastroenteritis worldwide. Noroviruses are responsible for up to 1.1 million hospitalizations and 218,000 deaths annually in children in the developing world.
There is currently no effective treatment or effective vaccine for norovirus, and the ability to curtail outbreaks is limited. We are developing a novel norovirus antiviral candidate for the prophylactic and therapeutic treatment of norovirus infection that is currently in a Phase 1 clinical study. A few companies have been developing vaccines and are in stages of clinical testing, including Vaxart Pharmaceutical, Moderna, Hillevax, Takeda Pharmaceuticals, Anhui Zhifei Longcom Biopharmaceutical (China) and National Vaccine and Serum Institute (China).
Coronavirus: COVID-19 continues to be a global pandemic fueled by an emergence of new strains
COVID-19 is a global health concern responsible for more than 777 million reported cases globally, including more than 7.1 million deaths, as of April 2026, according to data reported by the WHO.
Coronaviruses (CoV) are a large family of RNA viruses that historically have been associated with illness ranging from mild symptoms similar to the common cold to more severe respiratory disease. Infection with the novel SARS-CoV-2 has been associated with a wide range of responses, from no symptoms to more severe disease that has included pneumonia, severe acute respiratory syndrome, kidney failure, and death. The incubation period for SARS-CoV-2 is believed to be within 14 days after exposure, with most illness occurring within about five days after exposure. SARS-CoV-2, like other RNA viruses, is prone to mutate over time, resulting in the emergence of multiple variants. Adaptive mutations in the viral genome can alter the virus’s pathogenic potential. Even a single amino acid exchange can drastically affect a virus’s ability to evade the immune system and complicate the vaccine and antibody therapeutics development against the virus. Based on an epidemiological update by the WHO, five SARS-CoV-2 VOCs (variants of concern) have been identified since the beginning of the pandemic. Also, as demonstrated in the Delta, Omicron and other variants, some variations allow the virus to spread more easily and make it resistant to the treatments and vaccines.
| 6 |
On October 22, 2020, the U.S. Food and Drug Administration (“FDA”) approved the antiviral drug Veklury® (remdesivir) for the treatment of COVID-19 requiring hospitalization. Remdesivir is a nucleotide prodrug that inhibits viral replication and was previously evaluated in clinical trials for Ebola treatment in 2014. On May 25, 2023, the FDA approved Paxlovid™ (nirmatrelvir tablets and ritonavir tablets, co-packaged for oral use) for the treatment of mild-to-moderate COVID-19 in adults who are at high risk for progression to severe COVID-19, including hospitalization or death. For certain hospitalized adults with COVID-19, the FDA has also approved Olumiant® (baricitinib) and Actemra® (tocilizumab). In addition, the FDA issued emergency use authorization (EUA) for several antibody and antiviral therapeutics, including and Lagevrio™ (molnupiravir).
We continue pursuing the development of novel antiviral compounds for the treatment of coronavirus infections using our established proprietary drug discovery platform. By targeting the viral replication enzymes and protease, we believe it is possible to develop an effective treatment for all coronavirus diseases including COVID-19, Severe Acute Respiratory Syndrome (SARS), and Middle East Respiratory Syndrome (MERS).
Hepatitis C: A large competitive market with opportunity for shorter treatment regimens
HCV is a highly competitive and changing market. Since 2014, several combinations of direct-acting antiviral agents (“DAAs”) have been approved for the treatment of HCV infection. These include Harvoni® (sofosbuvir/ledipasvir) 12 weeks of treatment, Viekira Pak™ (ombitasvir/paritaprevir/ritonavir, dasabuvir) 12 weeks of treatment, Epclusa® (sofosbuvir/velpatasvir) 12 weeks of treatment, Zepatier™ (elbasvir/grazoprevir) 12 weeks of treatment and Mavyret® (glecaprevir/pibrentasvir) eight weeks of treatment. We believe the next improvements in HCV treatment will be ultra-short combination oral treatments of four to six weeks, which is the goal of our program.
We anticipate a significant global HCV market opportunity that will persist through at least 2036, given the large prevalence of HCV infection worldwide. The 2024 World Health Organization Global Hepatitis Report estimates that 50 million people worldwide have chronic HCV infections with about 1 million new infections occurring per year and an estimated 3.2 million adolescents and children with chronic HCV infection.
We are targeting the viral NS5B polymerase with a non-nucleoside inhibitor (“NNI”), which could be developed as part of an all-oral, pan-genotypic combination regimen. Our focus is on developing what is now called ultrashort treatment regimens from four to six weeks in length. Combining CC-31244 with different classes of approved DAAs has the potential to change the paradigm of treatment for HCV by shortening the duration of treatment. Combination strategies with approved drugs could allow us to expand CC-31244 into the HCV antiviral therapeutic area globally and could lead to a high and fast cure rate, to improved compliance, and to reduced treatment duration. To our knowledge no competing company has yet developed a short HCV treatment of less than 8 weeks with a high (>95%) sustained virologic response (SVR) at week 12.
CC-31244, an HCV NNI, is a potential best in class pan-genotypic inhibitor of NS5B polymerase for the treatment of HCV. We completed a randomized, double-blinded Phase 1a/b study in healthy volunteers and HCV-infected subjects in Canada in September 2016, with favorable safety results. We completed a Phase 2a study in HCV genotype 1 subjects in the U.S. in 2017. HCV-infected subjects treated with CC-31244 had a rapid and marked decline in HCV RNA levels, and slow viral rebound after treatment. Results of this study suggest that CC-31244 could be an important component in a shortened duration all-oral HCV combination therapy. In 2017, we completed the Phase 2a final study report as filed with the FDA.
We have been seeking a partner for further clinical development of CC-31244 since completing a Phase 2a study.
Results of Operations for the Three Months Ended March 31, 2026 compared to the Three Months Ended March 31, 2025
Grant Income
The Company received a $500,000 Small Business Innovation Research (“SBIR”) Phase I award from the National Institutes of Health (“NIH”) and the National Institute of Allergy and Infectious Diseases (NIAID). The NIH/NIAID Phase I award is designed to assess the scientific, technical and commercial potential of early-stage programs and will support the Company’s development of a novel, oral, broad-spectrum antiviral candidate for the treatment of influenza A and B infections.
Grant income is earned by award program progress from Company services provided. Total grant income for the three months ended March 31, 2026, and 2025 were $225,000 and $0, respectively.
Research and Development Expense
Research and development expense consists primarily of compensation-related costs for our employees dedicated to research and development activities and clinical trials, as well as lab supplies, lab services, and facilities and equipment costs related to our research and development programs.
Total research and development expenses for the three months ended March 31, 2026, and 2025 were $1,371,000 and $1,360,000, respectively.
| 7 |
General and Administrative Expense
General and administrative expenses include compensation-related costs for our employees dedicated to general and administrative activities, legal fees, audit and tax fees, consultants and professional services, and general corporate expenses.
General and administrative expenses for the three months ended March 31, 2026, and 2025 were $1,210,000 and $981,000, respectively. The increase of $229,000 was predominantly due to an increase in legal and consultant cost partially offset with a decrease of $76,000 on salary and wages.
Interest Income, Net
Interest income for the three months ended March 31, 2026 and 2025 was $22,000 and $37,000, respectively. The interest income was primarily earned on cash held in interest bearing bank accounts.
Other Income (Expense)
In 2022, the Company established a wholly owned subsidiary in Australia, making it subject to foreign exchange rate fluctuations. Foreign exchange gain of $35,000 and $3,000 was recorded for the three months ended March 31, 2026 and 2025, respectively.
Income Taxes
No income tax benefit or expense was recognized for the three months ended March 31, 2026 and 2025. The Company’s effective income tax rate was 0.00% for the three months ended March 31, 2026 and 2025. As a result of the Company’s cumulative losses, management has concluded that a full valuation allowance against the Company’s net deferred tax assets is appropriate.
Net Loss
As a result of the above factors, net loss for the three months ended March 31, 2026 was $2,299,000 compared with a net loss of $2,301,000 for the three months ended March 31, 2025, respectively, as a result of developments related to our three months ended March 31, 2026 grant income of $225,000 and expenses as described above.
Liquidity and Capital Resources
The Company’s consolidated financial statements are prepared using generally accepted accounting principles in the United States of America applicable to a going concern, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The Company has incurred net losses and negative operating cash flows since inception. For the three months ended March 31, 2026, the Company recorded a net loss of approximately $2,299,000 and used approximately $2,340,000 of cash in operating activities. These factors raise substantial doubt about the Company’s ability to continue as a going concern.
On March 31, 2026, the Company had cash and restricted cash of approximately $4,760,000. Restricted cash represents amounts pledged as collateral for financing arrangements that are currently limited to the issuance of business credit cards. The restriction will end upon the conclusion of these financing arrangements. We believe that our current resources will not be sufficient to fund our operations beyond the next 12 months. This estimate is based, in part, upon our currently projected expenditures. As a result, management has concluded that there is substantial doubt about the Company’s ability to continue as a going concern. The accompanying consolidated financial statements do not include any adjustments relating to the recoverability and classification of recorded assets, or the amounts and classification of liabilities that might be necessary in the event the company cannot continue as a going concern. The Company’s independent registered public accounting firm, in its report on the Company’s consolidated financial statements for the year ended December 31, 2025, has also expressed substantial doubt about the Company’s ability to continue as a going concern.
Net cash used in operating activities was $2,340,000 for the three months ended March 31, 2026 compared with net cash used in operating activities of $2,939,000 for the same period in 2025. This decrease was primarily due to decrease in period expenses related to our Influenza A Phase 2a clinical trial.
The Company has not yet established an ongoing source of revenue sufficient to cover its operating costs. The Company had $4,685,000 unrestricted cash on March 31, 2026. We expect that our reported cash balance is not be sufficient to support the Company’s working capital needs for the 12 months following the filing of this report, taking into account our intended research and development efforts for the remainder of 2026 and beyond.
Developing pharmaceutical products, including conducting preclinical studies and clinical trials, is capital-intensive. As a rule, research and development expenses increase substantially as a company advances a product candidate toward clinical programs. Historically, we have financed our operations with the proceeds from public and private equity and debt offerings, including additional investments by certain existing stockholders, and entered into strategic partnerships and collaborations for the research, development and commercialization of product candidates.
We have focused our efforts on research and development activities, including through collaborations with suitable partners. We have been profitable on a quarterly basis but have never been profitable on an annual basis. We have no products approved for sale and have incurred operating losses and negative operating cash flows on an annual basis since inception.
| 8 |
The Company’s interim consolidated financial statements are prepared using generally accepted accounting principles in the United States of America applicable to a going concern, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. Historically, public and private equity offerings have been our principal source of liquidity.
The Company is party to the At-The-Market Offering Agreement, dated July 1, 2020 (“ATM Agreement”) with H.C. Wainwright & Co., LLC (“Wainwright”), pursuant to which the Company may issue and sell over time and from time to time, to or through Wainwright, up to $10,000,000 of shares of the Company’s common stock. During January 2021, the Company sold 1,030,000 shares of its common stock pursuant to the ATM Agreement for net proceeds of approximately $2,072,000. On May 24, 2023, the Company filed a prospectus supplement covering sales under the ATM Agreement under which we may offer and sell shares of our common stock having an aggregate offering price of up to $7,250,000 from time to time through Wainwright. There were no sales under the ATM Agreement during the three months ended March 31, 2026.
As of the date of this Report, the Company has sold a total 1,200,152 shares of its common stock for total net proceeds of approximately $2,380,000 pursuant to the ATM Agreement. On September 12, 2025, the Company and Wainwright agreed to terminate the sales of shares under the ATM Agreement and the Company filed a prospectus supplement with the SEC to that effect. As a result of this, the at-the-market offering under the ATM Agreement is no longer ongoing as of September 12, 2025, and the Company will not make any sales of common stock pursuant to the ATM Agreement unless and until a new prospectus supplement is filed with the SEC; however, the ATM Agreement remains in full force and effect.
As the Company continues to incur losses, achieving profitability is dependent upon the successful development, approval and commercialization of its product candidates, and achieving a level of revenues adequate to support the Company’s cost structure. The Company may never achieve profitability, and unless and until it does, the Company will continue to need to raise additional capital. Management intends to fund future operations through additional private or public equity offerings and through arrangements with strategic partners or from other sources. There can be no assurances, however, that additional funding will be available on terms acceptable to the Company, or at all, and any equity financing may be very dilutive to existing stockholders.
Cautionary Note Regarding Forward-Looking Statements
This report includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including statements regarding the future effectiveness of our product candidates, our expectations regarding future characteristics of the product candidates we develop, our plans for the future development of preclinical and clinical drug candidates, the expected time of achieving certain value driving milestones in our programs and progressing our programs in the clinical development process generally, our expectations regarding future operating results and liquidity. The words “believe,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “could,” “target,” “potential,” “is likely,” “will,” “expect” and similar expressions, as they relate to us, are intended to identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs.
The results anticipated by any or all of these forward-looking statements might not occur. Important factors that could cause actual results to materially differ from those in the forward-looking statements include the risks and uncertainties arising from the risks arising from the possibility of a recession, interest rate increases, and the economic impact of United States tariff policies geopolitical conflicts including inflation, the wars in the Middle East and Ukraine on our Company, our collaboration partners, and on the U.S., U.K., Australia and global economies, including downturns in economic activity and capital markets, manufacturing and research delays arising from raw materials and labor shortages, supply chain disruptions and other business interruptions including any adverse impacts on our ability to obtain raw materials and test animals as well as similar problems with our vendors and our current and any future contract research organizations (CROs) and contract manufacturing organizations (CMOs), the progress and results of the studies for CC-42344 and CDI-988 including the delay of the Phase 2a study for CC-42344 which may require us to incur substantial additional costs, the results of the studies for CC-42344 and CDI-988 and any future preclinical and clinical trials, the ability of our CROs to recruit volunteers for, and to proceed with, clinical studies, and our collaboration partners’ technology and software performing as expected, financial difficulties experienced by certain partners, general risks arising from clinical trials, receipt of regulatory approvals and changes including based on initiatives and actions taken by the Trump Administration which could, among other things, result in delays in regulatory approvals or limit access to federal funding for our programs regulatory changes, development of effective treatments and/or vaccines by competitors, including as part of the programs financed by governmental authorities and potential mutations in a virus we are targeting which may result in variants that are resistant to a product candidate we develop. Further information on our risk factors is contained in our filings with the SEC, including our Annual Report on Form 10-K for the year ended December 31, 2025. We undertake no obligation to publicly update or revise any forward-looking statements, whether as the result of new information, future events or otherwise.
| 9 |
Critical Accounting Policies and Estimates
In our Annual Report on Form 10-K for the year ended December 31, 2025, we disclosed our critical accounting policies and estimates upon which our financial statements are derived.
Accounting estimates. The preparation of financial statements in conformity with accounting principles generally accepted in the U.S. requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ significantly from these estimates.
Readers are encouraged to review these disclosures in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025 in conjunction with the review of this report.
ITEM 3. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Not applicable.
ITEM 4. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
We carried out an evaluation, under the supervision and with the participation of our management, including our Co-Chief Executive Officers and Chief Financial Officer, of the effectiveness of our disclosure controls and procedures, as defined in Rules 13a-15(e) and 15d-15(e) of the Securities Exchange Act of 1934 (the “Exchange Act”) as of the end of the period covered by this report. Based on that evaluation, our Co-Chief Executive Officers and Chief Financial Officer have concluded that our disclosure controls and procedures as of March 31, 2026 were effective to ensure that information required to be disclosed by us in reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and forms.
Changes in Internal Control over Financial Reporting
There were no material changes in our internal controls over financial reporting or in other factors that could materially affect, or are reasonably likely to affect, our internal controls over financial reporting during the quarter ended March 31, 2026. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
| 10 |
PART II — OTHER INFORMATION
ITEM 1. LEGAL PROCEEDINGS
As disclosed in our Annual Report, a dispute has arisen with the United Kingdom clinical research organization (the “CRO”) that performed the Phase 2a study. The Company contends that the CRO breached its agreement and is requesting that the CRO refund the $6,309,000 it was paid or redo the study. The CRO has implicitly denied liability and is seeking to recover an additional approximately $600,000 from the Company. The Company has initiated arbitration to resolve the dispute as required under the agreement with the CRO. See the risk factor entitled “We face significant risks and uncertainties surrounding our Influenza A program following an initial Phase 2a study which failed to yield scientifically viable results relating to the product candidate’s efficacy” beginning on page 14 of our Annual Report and the discussion in Item 2 above under “Influenza Programs.” Additionally, from time to time, the Company is a party to, or otherwise involved in, legal proceedings arising in the normal course of business. During the reporting period, there have been no material changes to the description of legal proceedings set forth in our Annual Report on Form 10-K for the year ended December 31, 2025 and, .as of the date of this Report, the Company is not aware of any pending legal proceedings to which the Company or any of its subsidiaries is a party which, if determined adversely, would have a material effect on its business, results of operations, cash flows or financial position.
ITEM 1.A RISK FACTORS
None.
ITEM 2. UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDS
All recent sales of unregistered securities have been previously reported.
ITEM 3. DEFAULTS UPON SENIOR SECURITIES
None.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
ITEM 5. OTHER INFORMATION
During
the three months ended March 31, 2026, none of our directors or officers (as defined in Rule 16a-1(f) under the Exchange Act)
| 11 |
ITEM 6. EXHIBITS
The exhibits listed in the accompanying “Exhibit Index” are filed or incorporated by reference as part of this Form 10-Q.
EXHIBIT INDEX
| Exhibit | Incorporated by Reference | Filed or Furnished | ||||||||
| No. | Exhibit Description | Form | Date | Number | Herewith | |||||
| 3.1 | Certificate of Incorporation, as amended | 10-Q | 8/16/21 | 3.1 | ||||||
| 3.1(a) | Certificate of Amendment to Certificate of Incorporation – reverse stock split | 8-K | 10/3/22 | 3.1 | ||||||
| 3.1(b) | Certificate of Amendment to Certificate of Incorporation – reduce number of authorized shares | 8-K | 6/28/24 | 3.1 | ||||||
| 3.2 | Amended and Restated Bylaws | 8-K | 2/19/21 | 3.1 | ||||||
| 3.2(a) | Amendment No. 1 to Amended and Restated Bylaws | 8-K | 6/18/25 | 3.1 | ||||||
| 31.1 | Certification of Principal Executive Officer (302) | Filed | ||||||||
| 31.2 | Certification of Principal Executive Officer (302) | Filed | ||||||||
| 31.3 | Certification of Principal Financial Officer (302) | Filed | ||||||||
| 32.1 | Certification of Principal Executive and Principal Financial Officer (906) | Furnished* | ||||||||
| 101.INS | Inline XBRL Instance Document | Filed | ||||||||
| 101.SCH | Inline XBRL Taxonomy Extension Schema Document | Filed | ||||||||
| 101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase Document | Filed | ||||||||
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase Document | Filed | ||||||||
| 101.LAB | Inline XBRL Taxonomy Extension Label Linkbase Document | Filed | ||||||||
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase Document | Filed | ||||||||
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | Filed | ||||||||
* This exhibit is being furnished rather than filed and shall not be deemed incorporated by reference into any filing, in accordance with Item 601 of Regulation S-K.
Copies of this report (including the financial statements) and any of the exhibits referred to above will be furnished at no cost to our stockholders who make a written request to our Corporate Secretary at Cocrystal Pharma, Inc., 4400 Biscayne Blvd, Suite 101, Miami, FL 33137.
| 12 |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| Cocrystal Pharma, Inc. | ||
| Dated: May 15, 2026 | By: | /s/ Sam Lee |
| Sam Lee | ||
| President and Co-Chief Executive Officer | ||
| (Principal Executive Officer) | ||
| Dated: May 15, 2026 | By: | /s/ James Martin |
| James Martin | ||
| Chief Financial Officer and Co-Chief Executive Officer | ||
| (Principal Executive Officer and Principal Financial Officer) | ||
| 13 |